filmov
tv
Calculate the number of moles of gas present in the container of volume 10 L at 300 K. If the m...

Показать описание
Calculate the number of moles of gas present in the container of volume 10 L at 300 K. If the manometer containing glycerin shows 5 m difference in level as shown diagram.
(Given : d_glycerin =2.72 g / mL ; d_mercury =13.6 g / mL )
(a) 0.94 mole
(b) 0.49 mole
(c) 0.64 mole
(d) none of these
📌 PHYSICS WALLAH OTHER CHANNELS :
📌 PHYSICS WALLAH SOCIAL MEDIA PROFILES :
(Given : d_glycerin =2.72 g / mL ; d_mercury =13.6 g / mL )
(a) 0.94 mole
(b) 0.49 mole
(c) 0.64 mole
(d) none of these
📌 PHYSICS WALLAH OTHER CHANNELS :
📌 PHYSICS WALLAH SOCIAL MEDIA PROFILES :
 0:05:29
0:05:29
 0:04:29
0:04:29
 0:05:49
0:05:49
 0:11:46
0:11:46
 0:03:14
0:03:14
 0:03:16
0:03:16
 0:05:46
0:05:46
 0:06:06
0:06:06
 0:00:28
0:00:28
 0:06:04
0:06:04
 0:05:16
0:05:16
 0:02:57
0:02:57
 0:05:57
0:05:57
 0:05:59
0:05:59
 0:12:23
0:12:23
 0:08:06
0:08:06
 0:01:25
0:01:25
 0:06:40
0:06:40
 0:10:50
0:10:50
 0:13:17
0:13:17
 0:02:47
0:02:47
 0:06:12
0:06:12
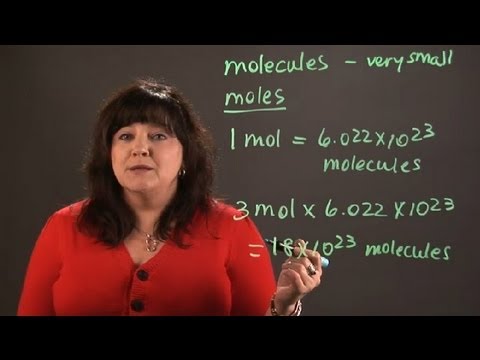 0:02:06
0:02:06
 0:12:11
0:12:11