filmov
tv
Calculate the wavelength, in nanometers,of the spectral line produced when an electron in a hydrogen

Показать описание
Calculate the wavelength, in nanometers, of the spectral line produced when an electron in a hydrogen atom undergoes the transition from the energy level 𝑛=3 to the level 𝑛=1.
solving for wavelength in nanometers
Calculate the wavelength, in nanometers,of the spectral line produced when an electron in a hydrogen
What wavelength in nanometers has a frequency of 8.6x10^13 Hz? - Frequency to Wavelength Practice
Speed of Light, Frequency, and Wavelength Calculations - Chemistry Practice Problems
How To Calculate The Energy of a Photon Given Frequency & Wavelength in nm Chemistry
Calculate the wavelength (in nanometer) associated with a proton mo...
Calculate the wavelength (in nanometers) of a photon emitted by a hydrogen atom when its electron d…...
Energy from Wavelength: Electromagnetic Radiation Calculation
Calculate the wavelength; in nanometers, of the light emitted by a hydrogen atom when its electron …...
Calculate the wavelength in nm of a x-ray wave of 2.0 × 1018 Hz | EM waves class 11 numerical
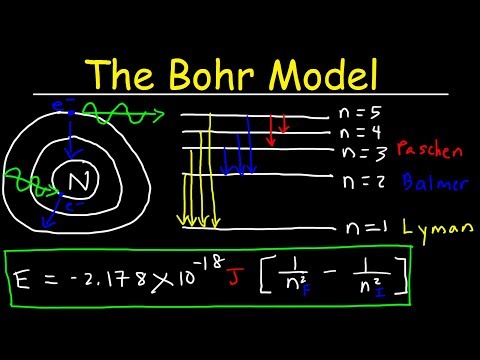
Bohr Model of the Hydrogen Atom, Electron Transitions, Atomic Energy Levels, Lyman & Balmer Seri...
Calculate the wavelength of the photon emitted when the hydrogen atom transition from n=5 to n=3.
How to Find the Wavelength, Frequency, Energy and Photons | Study Chemistry With Us
Frequency from Wavelength: Electromagnetic Radiation Calculation
Calculate the wavelength (in nm) associated with a proton moving at 1.0x103m/s. the mass of proton i
What is 624 nm in Joules? - Wavelength to Energy Practice
Calculate the wavelength (in nm) of the radiation with the minimum amount of energy required for th…...
Converting Frequency of light to wavelength (in nm)
Calculate the wavelength (in nm) of a photon emitted by a hydrogen atom when its electron drops fro…...
ALEKS: Calculating the wavelength of a line in the spectrum of hydrogen
Meters and Nanometers ⚛️ #chem #chemistrynotes #wavelength
The wavelength of the first line in the hbalmer series is `656 nm ` .Calculate the wavelength
Calculate the wavelength (in nanometer) associated with a proton moving at 1.0×10^3m/s.
What is 566 nm in Hertz? - Wavelength to Frequency Practice
Комментарии
 0:01:46
0:01:46
 0:03:35
0:03:35
 0:05:44
0:05:44
 0:11:36
0:11:36
 0:11:06
0:11:06
 0:02:15
0:02:15
 0:00:33
0:00:33
 0:04:43
0:04:43
 0:00:33
0:00:33
 0:02:01
0:02:01
 0:21:44
0:21:44
 0:02:54
0:02:54
 0:36:02
0:36:02
 0:05:35
0:05:35
 0:05:17
0:05:17
 0:07:52
0:07:52
 0:00:33
0:00:33
 0:02:29
0:02:29
 0:00:33
0:00:33
 0:07:25
0:07:25
 0:00:15
0:00:15
 0:02:57
0:02:57
 0:02:02
0:02:02
 0:06:16
0:06:16