filmov
tv
Ideal Gas Graph Sketching

Показать описание
In this video we want to learn how to use the Ideal Gas Equation to sketch graphs of an ideal gas.
Let's first take a look at the Ideal Gas Equation, PV = nRT
where:
P is pressure in Pa,
V is volume in m3,
n is moles of gas,
R is gas constant 8.31 J K-1 mol-1, and
T is temperature in K.
For graph sketching we have to use the Ideal Gas Equation and write the equation in terms of y = mx to work out the relationship between the x and y terms.
Let's have a few examples for a fixed mass of ideal gas.
1. P against V at constant T
First we need to find all the constant terms and combine them together.
Since mass is fixed and molar mass of an ideal gas is constant, number of mole, n, will be constant.
R and T are constants too therefore nRT is constant.
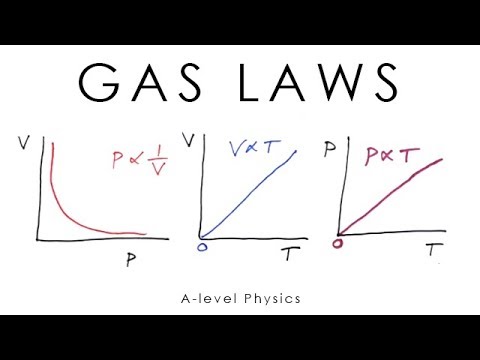
We can rearrange the equation where P is the y term and V is the x term to get the inverse relationship between P and V.
Therefore P against V will be an inverse graph.
2. P against 1/V at constant T
We already know P is inversely related to V so P is directly related to 1/V.
Therefore P against 1/V will be a straight line passing through origin, with gradient = k = nRT
3. PV against P at constant T
Again we know PV = nRT = k. Therefore PV will always be k regardless of the value of P.
This means PV against P will be a horizontal line which cuts the PV axis at k = nRT.
For the detailed step-by-step discussion on how to sketch the graph of an ideal gas using the Ideal Gas Equation, check out this video!
Topic: Gaseous State, Physical Chemistry, A Level Chemistry, Singapore
Found this video useful?
Please LIKE this video and SHARE it with your friends!
Any feedback, comments or questions to clarify?
Suggestions for new video lessons?
Drop them in the COMMENTS Section, I would love to hear from you!
Do check out the following for more video lessons:
-~-~~-~~~-~~-~-
Please watch my latest video: "2019 P1 Q2 - Deflection of Charged Particle in Electric Field"
-~-~~-~~~-~~-~-
Let's first take a look at the Ideal Gas Equation, PV = nRT
where:
P is pressure in Pa,
V is volume in m3,
n is moles of gas,
R is gas constant 8.31 J K-1 mol-1, and
T is temperature in K.
For graph sketching we have to use the Ideal Gas Equation and write the equation in terms of y = mx to work out the relationship between the x and y terms.
Let's have a few examples for a fixed mass of ideal gas.
1. P against V at constant T
First we need to find all the constant terms and combine them together.
Since mass is fixed and molar mass of an ideal gas is constant, number of mole, n, will be constant.
R and T are constants too therefore nRT is constant.
We can rearrange the equation where P is the y term and V is the x term to get the inverse relationship between P and V.
Therefore P against V will be an inverse graph.
2. P against 1/V at constant T
We already know P is inversely related to V so P is directly related to 1/V.
Therefore P against 1/V will be a straight line passing through origin, with gradient = k = nRT
3. PV against P at constant T
Again we know PV = nRT = k. Therefore PV will always be k regardless of the value of P.
This means PV against P will be a horizontal line which cuts the PV axis at k = nRT.
For the detailed step-by-step discussion on how to sketch the graph of an ideal gas using the Ideal Gas Equation, check out this video!
Topic: Gaseous State, Physical Chemistry, A Level Chemistry, Singapore
Found this video useful?
Please LIKE this video and SHARE it with your friends!
Any feedback, comments or questions to clarify?
Suggestions for new video lessons?
Drop them in the COMMENTS Section, I would love to hear from you!
Do check out the following for more video lessons:
-~-~~-~~~-~~-~-
Please watch my latest video: "2019 P1 Q2 - Deflection of Charged Particle in Electric Field"
-~-~~-~~~-~~-~-
Комментарии
 0:08:29
0:08:29
 0:04:05
0:04:05
 0:20:17
0:20:17
 0:00:15
0:00:15
 0:07:54
0:07:54
 0:02:41
0:02:41
 0:10:23
0:10:23
 0:07:53
0:07:53
 0:10:01
0:10:01
 0:24:17
0:24:17
 0:14:03
0:14:03
 0:05:28
0:05:28
 0:03:51
0:03:51
 0:13:24
0:13:24
 0:29:00
0:29:00
 0:23:31
0:23:31
 0:12:48
0:12:48
 0:01:47
0:01:47
 0:02:36
0:02:36
 0:25:13
0:25:13
 0:04:23
0:04:23
 0:05:01
0:05:01
 0:12:44
0:12:44
 0:02:08
0:02:08