filmov
tv
Thermodynamics - work done by polytropic process (Problem 4-7)

Показать описание
From Book: Thermodynamics An Engineering Approach 8th edition.
Q4-7: A piston-cylinder device initially contains 0.07 m3 of nitrogen gas at 130 kPa and 1808C. The nitrogen is now expanded to a pressure of 80 kPa polytropically with a polytropic exponent whose value is equal to the specific heat ratio (called isentropic expansion). Determine the final temperature and the boundary work done during this process.
For more videos Please like this video and subscribe.
Q4-7: A piston-cylinder device initially contains 0.07 m3 of nitrogen gas at 130 kPa and 1808C. The nitrogen is now expanded to a pressure of 80 kPa polytropically with a polytropic exponent whose value is equal to the specific heat ratio (called isentropic expansion). Determine the final temperature and the boundary work done during this process.
For more videos Please like this video and subscribe.
 0:05:44
0:05:44
 0:11:27
0:11:27
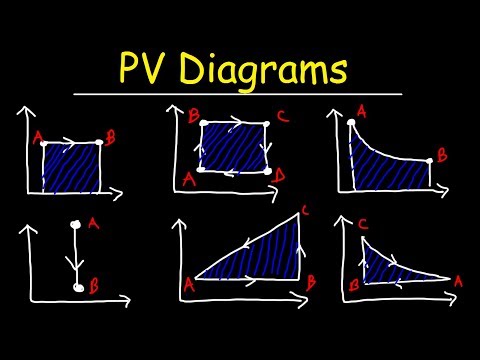 0:20:17
0:20:17
 0:04:39
0:04:39
 0:23:29
0:23:29
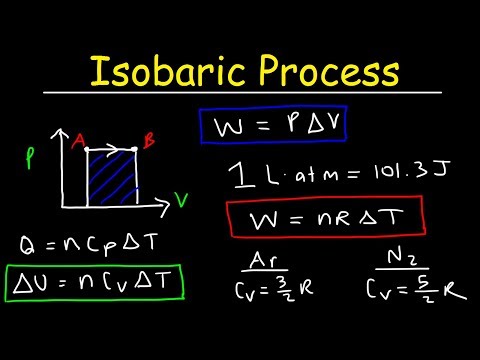 0:17:43
0:17:43
 0:10:15
0:10:15
 0:18:19
0:18:19
 0:00:55
0:00:55
 0:07:19
0:07:19
 0:16:42
0:16:42
 0:35:12
0:35:12
 0:11:43
0:11:43
 0:26:18
0:26:18
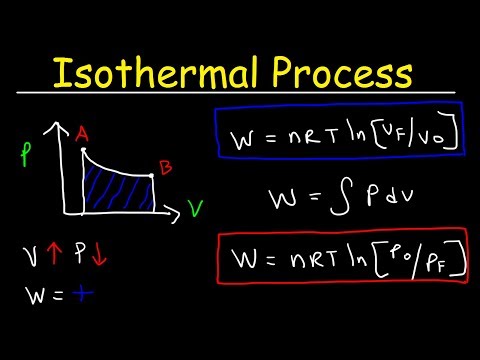 0:10:44
0:10:44
 0:07:10
0:07:10
 0:40:08
0:40:08
 0:04:23
0:04:23
 0:04:42
0:04:42
 0:16:41
0:16:41
 0:12:44
0:12:44
 0:16:45
0:16:45
 0:20:26
0:20:26
 0:08:48
0:08:48