filmov
tv
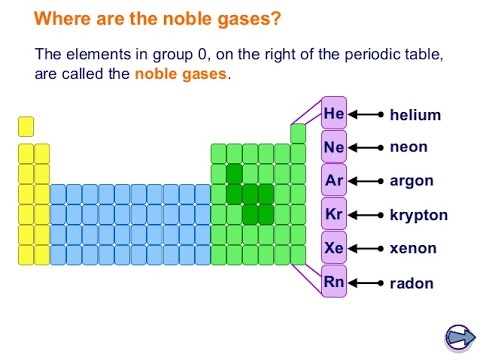
Elements of Group 0
Показать описание
Across
2. What state of matter are all Group 0 elements in at room temperature?
4. What word describes a chemical that is unreactive?
Down
1. Granite releases which radioactive gas?
3. Which group is also known as the noble gases?
Objectives
To understand that elements of Group 0 have similar properties.
To be able to recall the properties and uses of Group 0 elements.
To be able to explain why Group 0 elements are unreactive.
In your notes . . .
Use secondary sources of data to complete your poster on the noble gases.
Why are noble gases inert?
Noble gases are chemically unreactive or inert.
This is because they have a complete outer shell (highest energy level) of electrons.
Plenary
Match the noble gas to its use.
2. What state of matter are all Group 0 elements in at room temperature?
4. What word describes a chemical that is unreactive?
Down
1. Granite releases which radioactive gas?
3. Which group is also known as the noble gases?
Objectives
To understand that elements of Group 0 have similar properties.
To be able to recall the properties and uses of Group 0 elements.
To be able to explain why Group 0 elements are unreactive.
In your notes . . .
Use secondary sources of data to complete your poster on the noble gases.
Why are noble gases inert?
Noble gases are chemically unreactive or inert.
This is because they have a complete outer shell (highest energy level) of electrons.
Plenary
Match the noble gas to its use.
 0:04:49
0:04:49
 0:04:12
0:04:12
 0:05:46
0:05:46
 0:04:12
0:04:12
 0:00:11
0:00:11
 0:01:21
0:01:21
 0:06:24
0:06:24
 0:02:07
0:02:07
 0:23:32
0:23:32
 0:08:10
0:08:10
 0:00:46
0:00:46
 0:00:14
0:00:14
 0:05:36
0:05:36
 0:00:48
0:00:48
 0:06:07
0:06:07
 0:16:22
0:16:22
 0:06:12
0:06:12
 0:03:01
0:03:01
 0:03:30
0:03:30
 0:04:25
0:04:25
 0:02:34
0:02:34
 0:05:26
0:05:26
 0:02:34
0:02:34