filmov
tv
3.2. Condensed Phase Equilibrium

Показать описание
Lecture on the condensed-phase equilibrium of two- and three-component systems. For 2-component systems, the example system discussed is a binary system in which the components are completely miscible in the liquid phase. The application of the lever rule is also discussed. For 3-component systems, the Stokes-Rozeboom diagram is introduced with application to liquid-liquid-liquid systems and solid-solid-liquid systems.
OUTLINE
1:09 TWO-COMPONENT SYSTEMS (with complete miscibility in the liquid state)
2:17 Practical Applications
4:04 Variance of c=2 systems
5:45 CASE 1 - No chemical Reaction
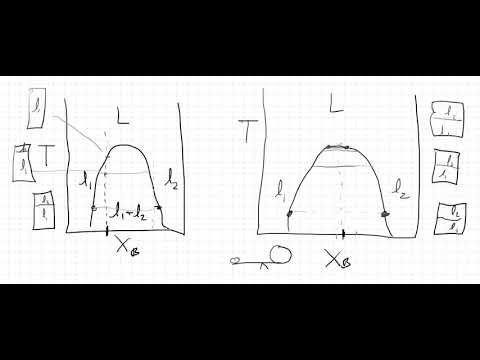
13:42 Tie lines - determining composition of a phase
15:47 The Lever Rule
39:29 CASE 2A - Eutectic reaction
45:43 CASE 2B - Peritectic reaction
57:55 THREE-COMPONENT SYSTEMS
59:55 The Stokes-Rozeboom Diagram
1:05:33 Liquid-liquid-liquid systems: analysis of differences in miscibility between the components
1:21:50 Solid-solid-liquid systems: no reaction between components
1:33:05 Solid-solid-liquid systems: pure components react to form a binary compound
OUTLINE
1:09 TWO-COMPONENT SYSTEMS (with complete miscibility in the liquid state)
2:17 Practical Applications
4:04 Variance of c=2 systems
5:45 CASE 1 - No chemical Reaction
13:42 Tie lines - determining composition of a phase
15:47 The Lever Rule
39:29 CASE 2A - Eutectic reaction
45:43 CASE 2B - Peritectic reaction
57:55 THREE-COMPONENT SYSTEMS
59:55 The Stokes-Rozeboom Diagram
1:05:33 Liquid-liquid-liquid systems: analysis of differences in miscibility between the components
1:21:50 Solid-solid-liquid systems: no reaction between components
1:33:05 Solid-solid-liquid systems: pure components react to form a binary compound
3.2. Condensed Phase Equilibrium
Phase Rule - Two component system
What is condensed phase rule? | Phase Rule | Physical Chemistry
10.1-Condensed Phases
Liquid-Liquid Phase Diagrams
3.1. Phase Equilibrium
26a Model of Condensed Phase Liquids Solids Pure
Equipartition Theorem - Condensed Phases
TWO COMPONENT SYSTEM - PHASE DIAGRAM -9 || PHASE RULE
Gibbs phase rule and Condensed phase rule
Three Phases (Temperature)
General Chemistry: Lec 5. Phase Diagrams and Phase Transitions
Lec 3 : Classical Thermodynamics of Phase Equilibria – 2
Condensed-Phase Spectroscopy
General Chemistry: Lec 3. Last Gas(p) and Condensed Phases
How much does a PHYSICS RESEARCHER make?
Eutectic Point | lead - Silver System | Condensed Phase Rule
Chemistry for Engineers - Condensed Phases–Solid
Phase Equilibria Diagram demonstration, Part 1
General Chemistry: Lec 4. Condensed Phases, Solids, & Phase Diagrams
Distillation
PHASE RULE || DEGREE OF FREEDOM || GIBBS PHASE RULE.
Condensed or reduced Phase Rule | Dr. Vibha Shrivastava | SISTec-E
Episode A7 - Thermodynamic Data for Condensed Mixtures
Комментарии
 1:47:08
1:47:08
 0:07:17
0:07:17
 0:01:49
0:01:49
 0:03:14
0:03:14
 0:11:51
0:11:51
 1:28:43
1:28:43
 0:29:10
0:29:10
 0:14:01
0:14:01
 0:14:50
0:14:50
 0:09:44
0:09:44
 0:10:05
0:10:05
 0:54:38
0:54:38
 1:00:02
1:00:02
 0:11:47
0:11:47
 1:05:20
1:05:20
 0:00:44
0:00:44
 0:16:18
0:16:18
 0:08:55
0:08:55
 0:04:08
0:04:08
 0:59:49
0:59:49
 0:10:58
0:10:58
 0:17:44
0:17:44
 0:02:23
0:02:23
 0:30:16
0:30:16