filmov
tv
⚗️ Calculating Density

Показать описание
Q. A man receives a ring he believes to be platinum from his fiancée. Before the wedding, he notices that the ring feels a little light for its size and decides to measure its density. He places the ring on a balance and finds that it has a mass of 3.15 grams. He then finds that the ring displaces 0.233 cm^3 of water.
Is the ring made of platinum?
(Note: The volume of irregularly shaped objects is often measured by the displacement of water. To use this method, the object is placed in water and the change in volume of the water is measured. The increase in the total volume represents the volume of water displaced by the object and is equal to the volume of the object.)
What you'll need:
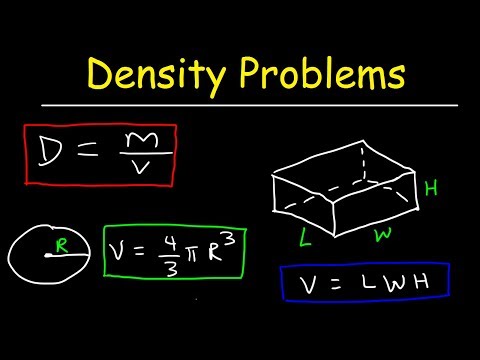
Relationship between density, mass, and volume: (d=m/V)
Density of platinum: (21.4 g/cm^3 )
 0:03:36
0:03:36
 0:06:47
0:06:47
 0:05:07
0:05:07
 0:18:35
0:18:35
 0:04:54
0:04:54
 0:07:55
0:07:55
 0:02:06
0:02:06
 0:05:17
0:05:17
 0:04:40
0:04:40
 0:03:40
0:03:40
 0:10:10
0:10:10
 0:12:20
0:12:20
 0:00:30
0:00:30
 0:05:04
0:05:04
 0:11:43
0:11:43
 0:06:09
0:06:09
 0:00:45
0:00:45
 0:03:10
0:03:10
 0:06:27
0:06:27
 0:00:56
0:00:56
 0:01:28
0:01:28
 0:38:29
0:38:29
 0:30:07
0:30:07
 0:00:16
0:00:16