filmov
tv
Complex Ion Formation

Показать описание
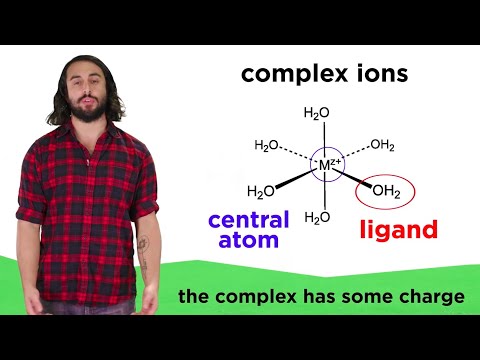
Most transition metal cations can do something interesting in solution, they can interact with specific ligands to form complex ions. These coordinate covalent bonds are new territory, so let's get a mini introduction to inorganic chemistry right here!
Check out "Is This Wi-Fi Organic?", my book on disarming pseudoscience!
Check out "Is This Wi-Fi Organic?", my book on disarming pseudoscience!
Complex Ion Formation
Complex Ion Equilibria - Stepwise Formation Constant Kf & Ksp Molar Solubility Problems
Complex Ions, Ligands, & Coordination Compounds, Basic Introduction Chemistry
Solubility and complex ion formation | Chemistry | Khan Academy
S3.1.8 - How are complex ions formed?
Complex Ion Formation | MCAT Organic Chemistry Prep
Ch 16 - Complex Ion Formation
Complex Ions and Their Ligands | A-level Chemistry | OCR, AQA, Edexcel
Metal pie complexes | Metal carbonyls | Easy explanation | Handwriiten notes❣️❣️
Solving a complex ion formation problem (Kf)
15.2 Complex Ion Formation
36 Formation of Complex Ions
S3.1.8 Complex ions (HL)
21.1 Introduction to Coordination Chemistry | General Chemistry
17.5.5 Complex Ion Formation and Coordination Complexes
Transition Element Complex Ion Formation from www.ChemistryTuition.Net
Complex ion formation affect on solubility
Ligands & complex ions for A level Chemistry
15 09A Complex Ion Formation
Transition metal complex ions
Complex Ion Formation
Complex ion formation
Complex-Ion Formation
Other Equilibrium Part 4, Complex ion formation
Комментарии
 0:04:06
0:04:06
 0:13:50
0:13:50
 0:13:42
0:13:42
 0:14:50
0:14:50
 0:05:01
0:05:01
 0:07:14
0:07:14
 0:14:11
0:14:11
 0:07:43
0:07:43
 0:09:41
0:09:41
 0:08:36
0:08:36
 0:32:46
0:32:46
 0:02:12
0:02:12
 0:04:22
0:04:22
 0:13:12
0:13:12
 0:07:33
0:07:33
 0:05:48
0:05:48
 0:11:44
0:11:44
 0:04:48
0:04:48
 0:08:09
0:08:09
 0:11:59
0:11:59
 0:10:59
0:10:59
 0:07:16
0:07:16
 0:28:10
0:28:10
 0:26:40
0:26:40